Introduction
Chimeric antigen receptor (CAR) T-cell therapy has revolutionized cancer treatment, particularly for B-cell malignancies. This innovative approach utilizes genetically engineered T cells equipped with synthetic receptors (CARs) to target and eliminate cancer cells. The effectiveness of CAR T-cell therapy hinges on the binding affinity of the CAR’s single-chain variable fragment (scFv) to specific antigens on cancer cells. M Car Tuning Bg refers to the process of modifying the scFv, also known as the binder, to optimize CAR T-cell performance. This article explores the structural mechanisms of two clinically relevant CD19 binders, FMC63 and SJ25C1, and how manipulating their binding affinities impacts CAR T-cell functionality, contributing to advancements in m car tuning bg.
Understanding CD19 Binder Engagement
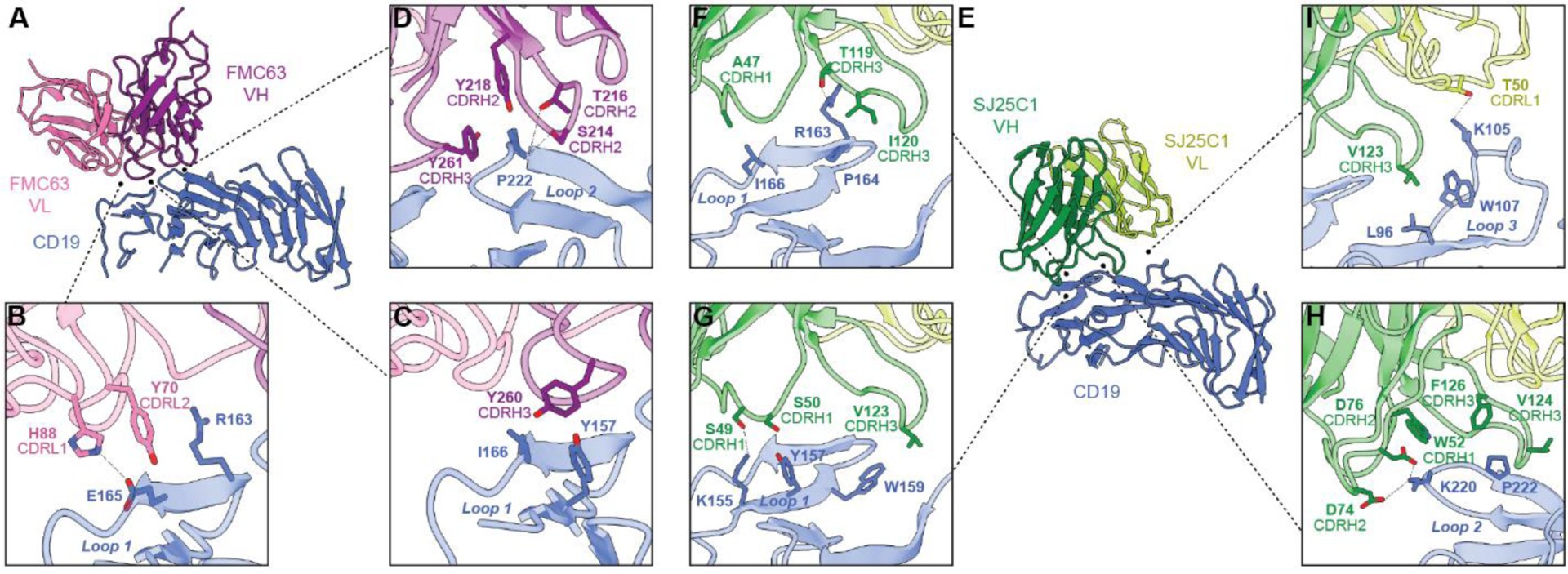
Cryo-electron microscopy (cryo-EM) structures of FMC63 and SJ25C1 in complex with the CD19 antigen reveal crucial insights into their binding mechanisms. FMC63, utilized in four FDA-approved CAR T-cell therapies, predominantly interacts with CD19 through its heavy variable (VH) domain, specifically targeting loops 1 and 2 on the antigen’s surface. SJ25C1, also extensively used in clinical trials, engages CD19 with a different orientation, primarily involving its VH domain and contacting loops 1, 2, and 3. Notably, both binders converge on a similar antigenic patch on CD19, comprised of these three loops.
M Car Tuning BG: Affinity Tuning for Optimized Performance
Molecular dynamics (MD) simulations coupled with Molecular Mechanics-Generalized Born Surface Area (MM/GBSA) analysis identified key residues contributing significantly to the binding energy between the binders and CD19. This information guided the design of mutant binders with altered affinities. Substitutions of tyrosine residues in FMC63 (Y70A, Y260A, Y261A) and isoleucine, valine, and tryptophan residues in SJ25C1 (I120A, I120V, I120G, V123G, V124G, W52F) were created and their binding affinities measured using surface plasmon resonance (SPR).
Impact of Affinity Tuning on CAR T-Cell Function
CAR T cells incorporating the engineered binders were evaluated for their cytolytic activity, antigen binding capacity, trogocytosis (antigen uptake), and cytokine release. Lowering the affinity of FMC63 and SJ25C1 resulted in CAR T cells with varying sensitivities to CD19 antigen density on target cells. Mutants with weaker affinities demonstrated reduced cytolytic activity against cells with low CD19 expression while maintaining effectiveness against high-density targets. Furthermore, affinity tuning influenced trogocytosis, a process implicated in CAR T-cell fratricide. Lower affinity binders exhibited decreased trogocytosis, potentially enhancing CAR T-cell persistence.
Conclusion
This research demonstrates the profound impact of m car tuning bg on CAR T-cell therapy. By leveraging structural biology, computational modeling, and biochemical analysis, researchers can fine-tune binder affinity to optimize CAR T-cell performance. Engineering lower affinity binders allows for the development of CAR T cells that selectively target cancer cells with high antigen density, potentially minimizing off-target effects. Moreover, modulating trogocytosis through affinity tuning offers a promising strategy to improve CAR T-cell persistence and overall therapeutic efficacy. This structure-guided approach to CAR engineering holds significant promise for advancing cancer immunotherapy and highlights the importance of continued research in m car tuning bg. The data and code used in this research are available as indicated in the original article.